What if the secret to living past 115 isn’t just good genes, but a precise biological harmony that keeps your body young even as time marches on? A groundbreaking multiomics study of the world’s oldest verified person – Maria Brañas Morera (or M116), a woman who lived to 117 years and 168 days – offers unprecedented insights into how extreme longevity and health can coexist, even in the face of molecular aging.
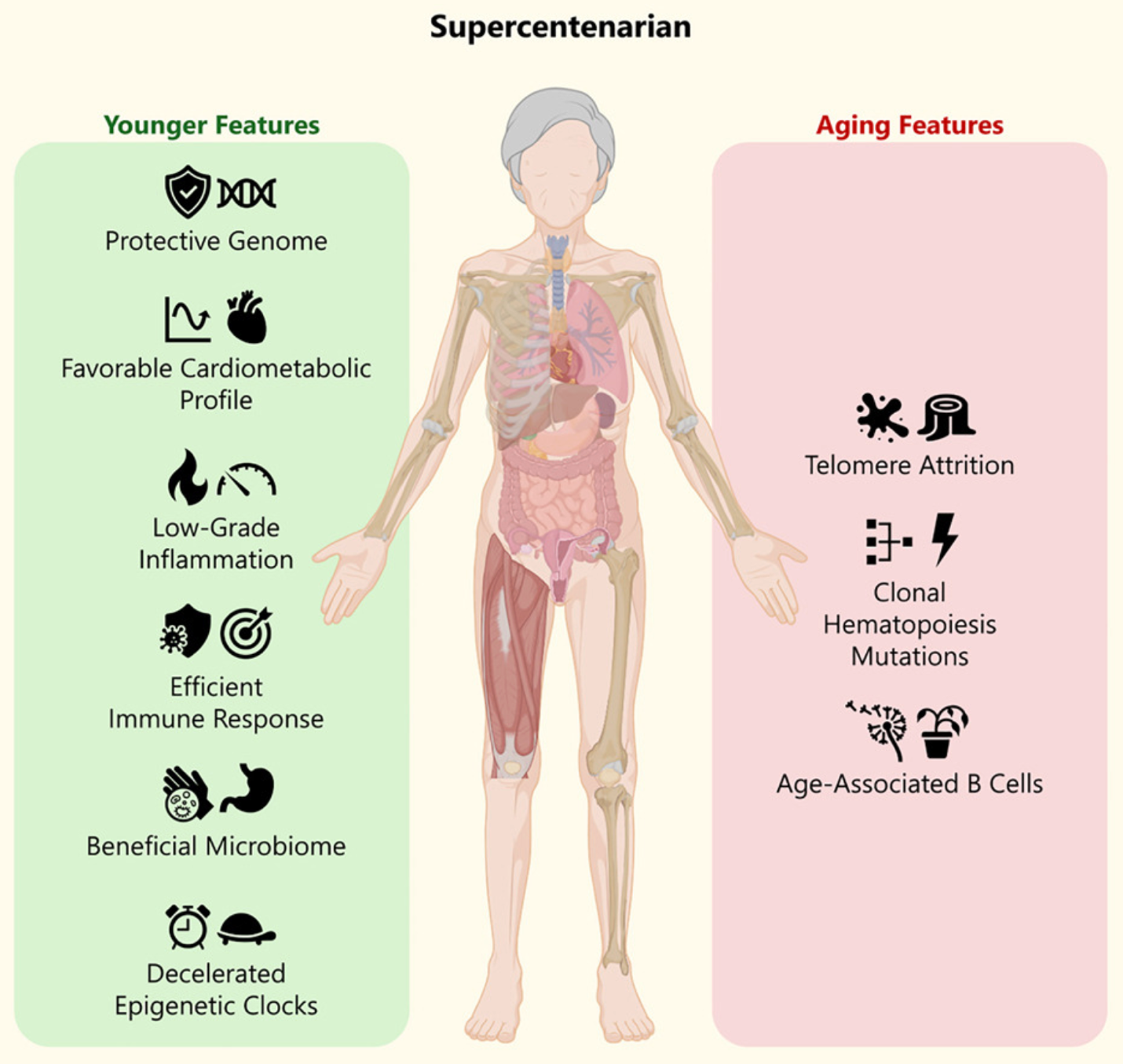
Published in Cell Reports Medicine, this research analyzed M116’s genome, epigenome, transcriptome, proteome, metabolome, and microbiome. The findings reveal a paradox: while her cells bore classic hallmarks of advanced age, like critically short telomeres and clonal blood mutations, her biological systems functioned with remarkable youthfulness. This duality challenges the long-held assumption that aging and disease are inseparable.
Published in Cell Reports Medicine, this research analyzed M116’s genome, epigenome, transcriptome, proteome, metabolome, and microbiome. The findings reveal a paradox: while her cells bore classic hallmarks of advanced age, like critically short telomeres and clonal blood mutations, her biological systems functioned with remarkable youthfulness. This duality challenges the long-held assumption that aging and disease are inseparable.
“Old” Body with a “Young” Biology
M116’s telomeres – the protective caps at chromosome ends – were among the shortest ever recorded in healthy individuals, averaging just 8 kilobases. Normally, such erosion predicts high risk of cancer or neurodegeneration. Yet M116 never developed Alzheimer’s, cancer, or cardiovascular disease. Instead, her body seemed to treat telomere shortening as a chronological clock rather than a pathological trigger.
Even more striking: despite carrying mutations in TET2 and SF3B1 (genes linked to age-related clonal hematopoiesis and blood cancer) she remained cancer-free. This suggests her immune system or other protective mechanisms successfully contained potential malignancies.
Even more striking: despite carrying mutations in TET2 and SF3B1 (genes linked to age-related clonal hematopoiesis and blood cancer) she remained cancer-free. This suggests her immune system or other protective mechanisms successfully contained potential malignancies.
A Genetic Blueprint for Resilience
Whole-genome sequencing uncovered rare, likely protective variants absent in control populations. Notably, she carried homozygous variants in genes tied to immune regulation (DSCAML1), heart health (PCDHA cluster), and mitochondrial efficiency (ND5, COX1). Her mitochondrial function, tested in live blood cells, was not just preserved – it was more robust than in younger controls, with higher membrane potential and energy output.
She also lacked high-risk alleles like APOE-ε4 (linked to Alzheimer’s) and instead carried longevity-associated variants in RABGAP1L and SOX6. Rather than one “longevity gene,” her genome revealed a network of rare, synergistic variants across immunity, metabolism, and neural protection.
She also lacked high-risk alleles like APOE-ε4 (linked to Alzheimer’s) and instead carried longevity-associated variants in RABGAP1L and SOX6. Rather than one “longevity gene,” her genome revealed a network of rare, synergistic variants across immunity, metabolism, and neural protection.
Youthful Signals in Blood and Gut
Her plasma told another story of resilience. Metabolomic profiling showed an exceptionally efficient lipid metabolism: very low triglycerides and VLDL, high HDL (“good cholesterol”), and abundant large HDL particles – patterns strongly associated with extended lifespan in UK Biobank data. Inflammation markers like GlycA and GlycB were remarkably low, indicating minimal systemic inflammation despite her age.
Her gut microbiome defied typical aging trends. While most elderly individuals lose microbial diversity, M116’s stool samples showed high α-diversity and an abundance of Bifidobacterium – a beneficial genus known for anti-inflammatory effects and short-chain fatty acid production. This profile resembles that of younger, healthier adults and aligns with her lifelong Mediterranean diet and daily yogurt consumption.
Her gut microbiome defied typical aging trends. While most elderly individuals lose microbial diversity, M116’s stool samples showed high α-diversity and an abundance of Bifidobacterium – a beneficial genus known for anti-inflammatory effects and short-chain fatty acid production. This profile resembles that of younger, healthier adults and aligns with her lifelong Mediterranean diet and daily yogurt consumption.
Epigenetic Youth
Perhaps the most compelling finding came from epigenetic clocks – algorithms that estimate biological age from DNA methylation patterns. Across six different clocks, M116’s blood, saliva, and urine consistently showed a biological age 23 years younger than her chronological age. Even a completely independent clock based on ribosomal DNA methylation confirmed this deceleration.
This epigenetic youthfulness wasn’t random. Her repetitive DNA elements (like LINE-1 and ALU), which usually lose methylation with age, remained tightly silenced – likely preventing genomic instability. Meanwhile, key regulatory genes showed methylation patterns linked to vascular health, metabolism, and bone integrity.
This epigenetic youthfulness wasn’t random. Her repetitive DNA elements (like LINE-1 and ALU), which usually lose methylation with age, remained tightly silenced – likely preventing genomic instability. Meanwhile, key regulatory genes showed methylation patterns linked to vascular health, metabolism, and bone integrity.
Practical Lessons for Healthy Aging
M116’s life wasn’t just about genes. She followed a Mediterranean diet, stayed socially engaged, walked regularly, and played piano into her final years. Her high yogurt intake may have nurtured her Bifidobacterium-rich gut, which in turn supported low inflammation and metabolic health. This synergy between lifestyle and biology underscores a key message: longevity isn’t predetermined – it’s modifiable.
Caveats and Future Directions
The study’s biggest limitation is its sample size: one person. While M116’s data are richly contextualized against large control cohorts, individual variability in aging is immense. Her unique genetic background – Spanish descent with lifelong residence in Catalonia – also limits generalizability.
Moreover, we can’t prove causality. Did her yogurt cause her Bifidobacterium bloom? Did her rare variants directly prevent disease? These questions require longitudinal studies in diverse supercentenarian cohorts.
Still, this research shifts the paradigm. Aging isn’t a monolithic decline but a mosaic of processes – some deteriorating, others resilient. The goal of longevity science may not be to stop aging entirely, but to decouple it from disease, as M116 so elegantly did.
Moreover, we can’t prove causality. Did her yogurt cause her Bifidobacterium bloom? Did her rare variants directly prevent disease? These questions require longitudinal studies in diverse supercentenarian cohorts.
Still, this research shifts the paradigm. Aging isn’t a monolithic decline but a mosaic of processes – some deteriorating, others resilient. The goal of longevity science may not be to stop aging entirely, but to decouple it from disease, as M116 so elegantly did.
Conclusion: A New Vision of Longevity
M116’s legacy is more than a record. She embodies a biological ideal: extreme lifespan without the burden of age-related disease. Her multiomics profile offers a roadmap – not of a single miracle drug, but of interconnected systems working in harmony. As researchers work to translate these insights into therapies, one truth emerges: the future of aging isn’t just about living longer, but about living better, right to the very end.
Publication date: 16.10.2025
Source:
Santos-Pujol E. et al. The multiomics blueprint of the individual with the most extreme lifespan. Cell Rep Med. 2025 Sep 24:102368. doi: 10.1016/j.xcrm.2025.102368. Epub ahead of print. PMID: 40997805.
https://www.cell.com/cell-reports-medicine/fulltext/S2666-3791(25)00441-0
Santos-Pujol E. et al. The multiomics blueprint of the individual with the most extreme lifespan. Cell Rep Med. 2025 Sep 24:102368. doi: 10.1016/j.xcrm.2025.102368. Epub ahead of print. PMID: 40997805.
https://www.cell.com/cell-reports-medicine/fulltext/S2666-3791(25)00441-0